Adsorption for Gas Separation from Combustion Sources
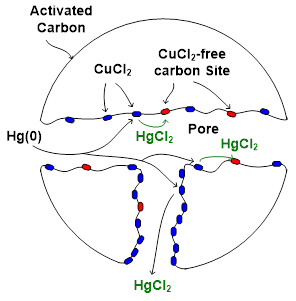
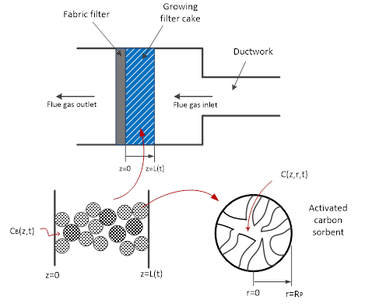
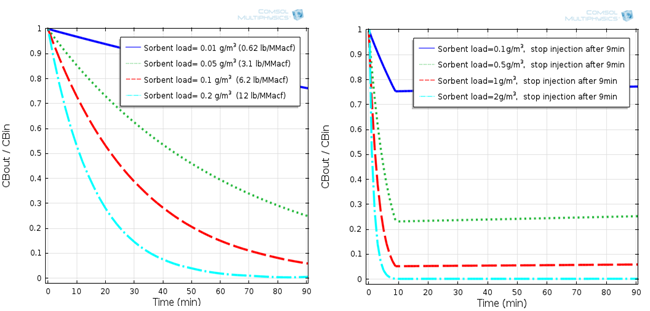
Our lab also develops adsorbents for gas separation in energy and environmental applications. We found that CuCl2-impregnated activated carbon (AC) sorbent captures Hg(0) by oxidizing it to mercuric chloride (HgCl2) followed by physical adsorption. Therefore, we studied mercuric chloride (HgCl2) adsorption onto activated carbon (AC) sorbent by separating the physical adsorption part to determine the adsorption kinetics. Based on the adsorption kinetics, we formulated a comprehensive model to predict HgCl2 removal in the ductwork and fabric filter by AC sorbent injection. We also studied various factors that could impact the adsorption performance and sorbent utilization, including inlet HgCl2 concentration, sorbent loading, particle size, external and pore diffusional mass-transfer resistances, residence time, filtration time, injection mode, and pressure drop. Throughout our adsorption kinetic and modeling study, we demonstrated that for the removal in fabric filter, a discontinuous sorbent injection mode delivering the same amount of sorbent in 10% of a cleaning cycle resulted in higher removal performance and sorbent utilization. However, at the end of the cleaning cycle, most of the sorbent capacity was not used (< 0.2%). The pressure drop across the filter cake built by fly ash and sorbent was found to be manageable within typical operating limits. Our studies in this area can be found in Publications.