Environmental Catalysis for Separation of Air Pollutants from Combustion Sources
Among many air pollutants in combustion flue gas, our lab studies the reaction and separation of mercury and CO2 gases using catalysis, adsorption, and absorption. Mercury is a toxic and persistent pollutant that bio-accumulates in the food chain, and most Americans are exposed to mercury primarily by consuming contaminated fish where mercury in the environment is transformed into more toxic methylmercury. The consumption of methylmercury may cause neurotoxic effects, and children and particularly developing fetuses are especially susceptible to methylmercury effects. The US EPA’s Mercury and Air Toxics Standards rule requires coal- and oil-fired power plants to use maximum available control technology to reduce the emissions of mercury and other hazardous air pollutants. Among elemental, oxidized, and particulate-bound mercury species present in flue gas, elemental mercury (Hg(0)) is most difficult to control because of its low concentrations, low reactivity with other flue gas components via homogenous oxidation, and low solubility in water. Therefore, the heterogeneous oxidation of Hg(0) vapor is required for separation of mercury in downstream air pollution control devices.
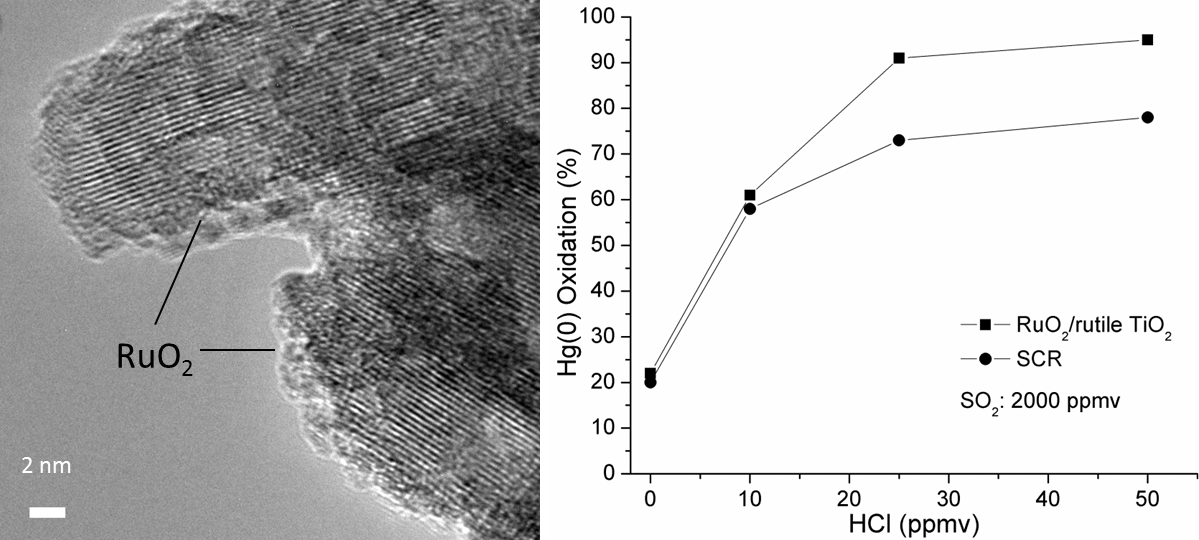
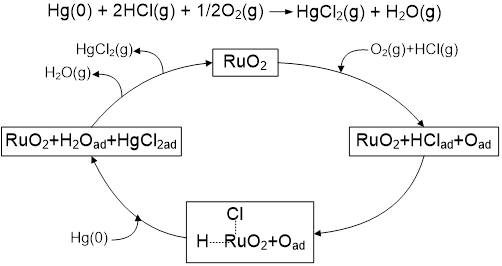
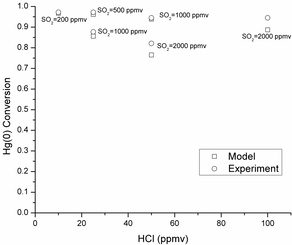
We aim to develop high- and low-temperature catalysts that can be used in an existing Selective Catalytic Reduction (SCR) unit and downstream after an air preheater in combustion process. We studied a high-temperature RuO2 catalyst to be used in a SCR unit at 300-400 °C. When we used rutile TiO2 as a catalyst support, RuO2 formed well dispersed nano-layers, giving higher Hg(0) oxidation activity over anatase TiO2 support. The RuO2/rutile TiO2 catalyst can be used at the tail end section of the selective catalytic reduction (SCR) unit for Hg(0) oxidation. It showed excellent Hg(0) oxidation performance under sub-bituminous and lignite coal simulated flue gas conditions with low concentration of HCl or HBr gas. The RuO2/rutile TiO2 catalyst also showed excellent resistance to SO2 under bituminous coal simulated flue gas, maintaining greater than 90% Hg(0) oxidation with ~2,000 ppmv SO2 present. The oxidized mercury in the form of HgCl2 has a high solubility in water and can be easily be separated in wet flue gas desulfurization scrubbers. We systematically studied the RuO2/rutile TiO2 catalyst for Hg(0) vapor oxidation in terms of synthesis, reaction mechanisms and kinetics. Our studies in this area can be found in Publications.
We aim to develop high- and low-temperature catalysts that can be used in an existing Selective Catalytic Reduction (SCR) unit and downstream after an air preheater in combustion process. We studied a high-temperature RuO2 catalyst to be used in a SCR unit at 300-400 °C. When we used rutile TiO2 as a catalyst support, RuO2 formed well dispersed nano-layers, giving higher Hg(0) oxidation activity over anatase TiO2 support. The RuO2/rutile TiO2 catalyst can be used at the tail end section of the selective catalytic reduction (SCR) unit for Hg(0) oxidation. It showed excellent Hg(0) oxidation performance under sub-bituminous and lignite coal simulated flue gas conditions with low concentration of HCl or HBr gas. The RuO2/rutile TiO2 catalyst also showed excellent resistance to SO2 under bituminous coal simulated flue gas, maintaining greater than 90% Hg(0) oxidation with ~2,000 ppmv SO2 present. The oxidized mercury in the form of HgCl2 has a high solubility in water and can be easily be separated in wet flue gas desulfurization scrubbers. We systematically studied the RuO2/rutile TiO2 catalyst for Hg(0) vapor oxidation in terms of synthesis, reaction mechanisms and kinetics. Our studies in this area can be found in Publications.
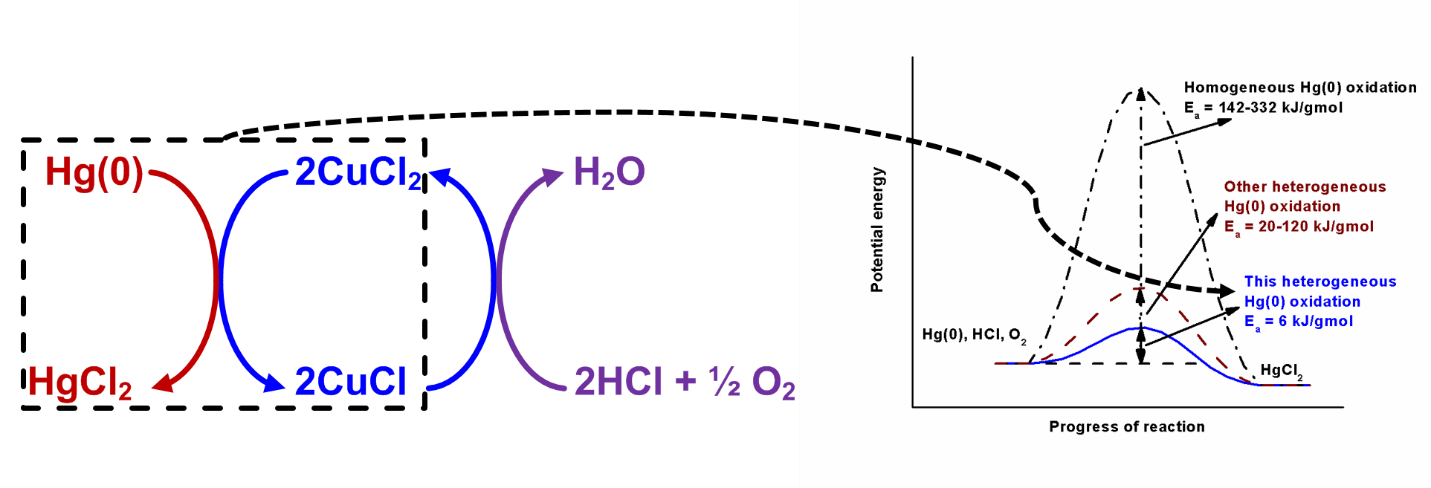
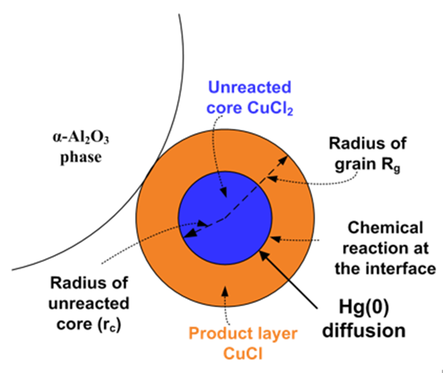
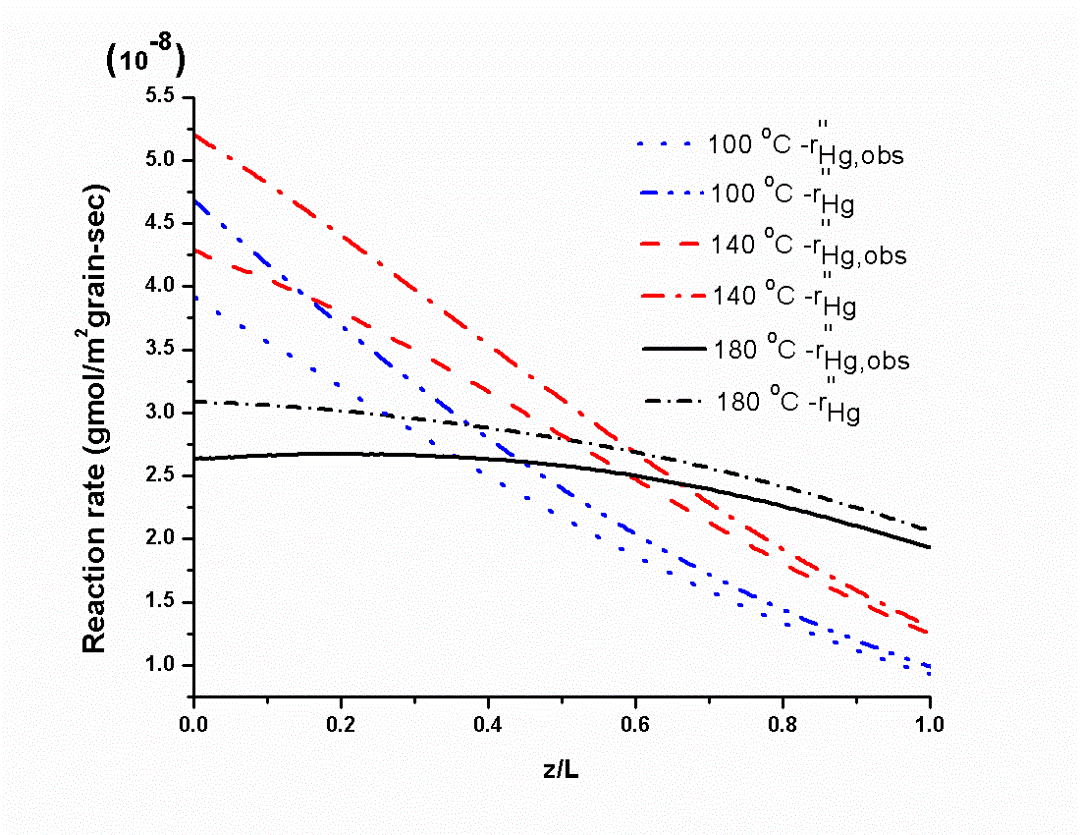
We also study the reaction mechanism and kinetics of a CuCl2-based low-temperature Hg(0) oxidation catalyst for plants without SCR unit in a temperature window of ~50-200 °C (~120-400 °F). We found CuCl2 to work as an excellent redox catalyst for heterogeneous Hg(0) oxidation by reducing itself to CuCl and being reoxidized back to CuCl2 with HCl and O2 gases present in typical coal combustion flue gas. Unlike many metal oxide-based catalysts, CuCl2 also showed excellent resistance to SO2 for Hg(0) vapor oxidation. We conducted a mechanistic study for Hg(0) oxidation with CuCl2/g-Al2O3 catalyst with high surface area. We found that CuCl2 formed agglomerates with an increase in temperature leading to sintering effect. Our reaction kinetic study using unsteady-state grain model showed that CuCl2 could enhance Hg(0) oxidation by lowering the activation energy barrier with the reduction of Cu(2+) to Cu(1+) and supplying thermally stable surface Cl sites following a Mars-Maessen mechanism. Our studies in this area can be found in (paper links).